【文献名】
Stuart J. Connolly, M.D. Dabigatran versus Warfarin in Patients with Atrial Fibrillation. N Engl J Med 2009; 361:1139-1151
【要約】
<Background>
Warfarin reduces the risk of stroke in patients with atrial fibrillation but increases the risk of hemorrhage and is difficult to use. Dabigatran is a new oral direct thrombin inhibitor.
<Methods>
In this noninferiority trial, we randomly assigned 18,113 patients who had atrial fibrillation and a risk of stroke to receive, in a blinded fashion, fixed doses of dabigatran ? 110 mg or 150 mg twice daily ? or, in an unblinded fashion, adjusted-dose warfarin. The median duration of the follow-up period was 2.0 years. The primary study outcome was stroke or systemic embolism.
Systemic embolism was defined as an acute vascular occlusion of an extremity or organ,documented by means of imaging,surgery,or autopsy.
The primary safety outcome was major hemorrhage.
<Results>
Characteristics of the study patients was described table 1 in the original article. The three treatment groups were well balanced with respect to baseline characteristics
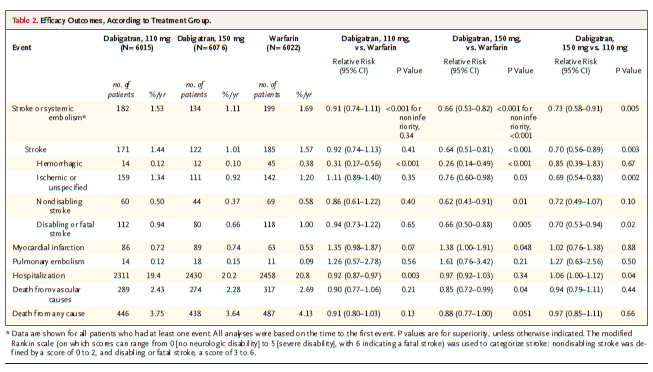
Rates of the primary outcome were 1.69% per year in the warfarin group, as compared with 1.53% per year in the group that received 110 mg of dabigatran (relative risk with dabigatran, 0.91; 95% CI, 0.74 to 1.11; P<0.001 for noninferiority) and 1.11% per year in the group that received 150 mg of dabigatran (relative risk, 0.66; 95% CI, 0.53 to 0.82; P<0.001 for superiority).
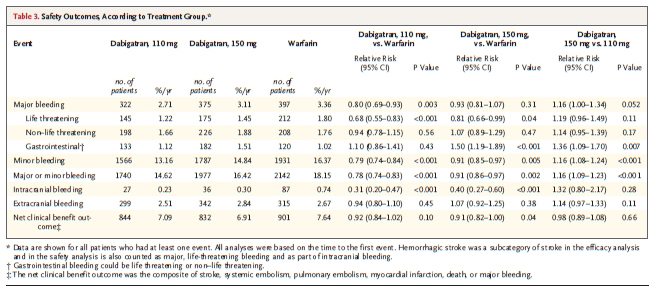
The rate of major bleeding was 3.36% per year in the warfarin group, as compared with 2.71% per year in the group receiving 110 mg of dabigatran (P=0.003) and 3.11% per year in the group receiving 150 mg of dabigatran (P=0.31). The rate of hemorrhagic stroke was 0.38% per year in the warfarin group, as compared with 0.12% per year with 110 mg of dabigatran (P<0.001) and 0.10% per year with 150 mg of dabigatran (P<0.001). The mortality rate was 4.13% per year in the warfarin group, as compared with 3.75% per year with 110 mg of dabigatran (P=0.13) and 3.64% per year with 150 mg of dabigatran (P=0.051).
The only adverse effect that was significantly more common with dabigatran than with warfarin was dyspepsia(Table.4)Dyspepsia occurred in 348 patients (5.8%) in the warfarin group and in 707 patients (11.8%) and 688 patients (11.3%) in the 110-mg and 150-mg dabigatran groups, respectively (P<0.001 for both comparisons)
<Conclusions>
In patients with atrial fibrillation, dabigatran given at a dose of 110 mg was associated with rates of stroke and systemic embolism that were similar to those associated with warfarin, as well as lower rates of major hemorrhage. Dabigatran administered at a dose of 150 mg, as compared with warfarin, was associated with lower rates of stroke and systemic embolism but similar rates of major hemorrhage.
【開催日】
2011年6月22日